|
Then we would wind up with X, which is the Hydro Nehemiah concentration equal to the square root of Casey and the hydro name ion. Well, I don't think so, but let's calculated and see if we wrote out the standard equilibrium and made the assumption that X would be small. Um, this one, huh? Wonder about that is the hydro Nia, my on 0.1. So we're not gonna wind up with, um, Mawr than 0.1.Ĭan't store geometrically wind up with more than 0.1 of the hydrogen sulfide ions. Now, uh, that first Proton is not gonna be strong acid. And we've got five choices for what would be true here. So we have a, uh, sulfurous acid solution at 0.1 Moeller and we are given the two k is for the two protons associations. So we have six times zero squared which is just zero, 16 times zero is again just zero. So we're going to do is plug in zero for X into this equation that we found in part B. And for part see what we're gonna do is we're gonna find the value of K. Which is an equal to six X squared minus 16 X. So we have six X squared minus 16 X plus four minus three. And here I'll simplify because it's a little bit easier. So we're gonna have two times three X squared -8 x plus two. So if we go back and look at our KFX equation, it's just two X -3. So in this case we plug in H of X for every value of X. And for part B, what we're going to figure out is the composite function K. I'm just going to leave it in this form as it is our composite function each of kfx. 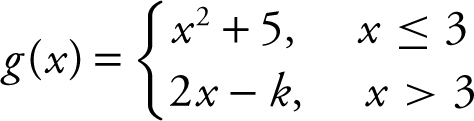
And then plus two.Īnd if you want you can actually expand this and then combine like terms and simplify a little bit. 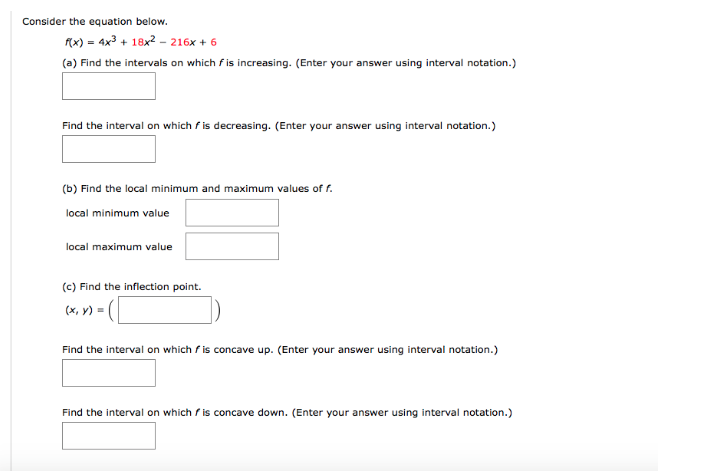
So you have three times two X minus three squared minus eight times two X minus three. So if we do this we'd plug in two x minus three into this X squared. And so the way that we do this as we just plug in cave X. 
And in part a what we're going to find is the composite function each of K.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
